Roger Greenhalgh (top left), Gunnar Tepe (top right), Jean-Paul de Vries (bottom left), Andrew Holden (bottom middle) and Amer Zanabili (bottom right)
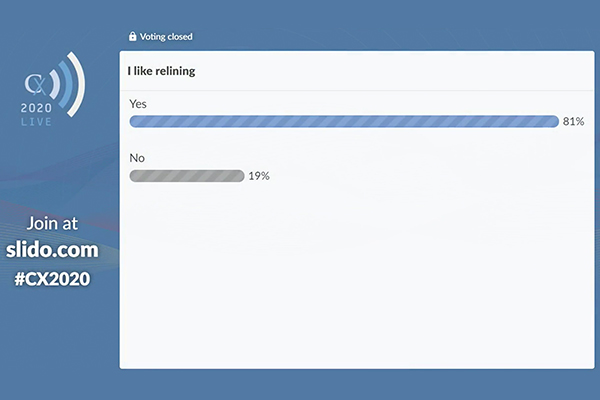
An overwhelming majority of 81% answered “yes” in agreement with the comment “I like relining”. The poll followed three sessions that looked at the concept of “relining”, using covered stents for the management of complex aortoiliac occlusive disease. Livestream attendees heard that the surgical management of complex lesions, which may be heavily calcified, is difficult, and may lead to rupture. Meanwhile, the endovascular approach is gaining ground. Amer Zanabili (Oviedo, Spain) presented an edited case demonstrating the relining of an occluded, long TASC D iliac lesion, showcasing the potential of this technique. The polling results that followed then revealed an enthusiasm for this endovascular approach.
Roger Greenhalgh (London, UK) chaired the discussion, and Gunnar Tepe (Rosenheim, Germany) joined him as moderator for the session.
Covered balloon-expandable stents are effective treatment options for aortoiliac occlusive disease
 In the first talk, Jean-Paul de Vries, (Groningen, The Netherlands) reported the key findings from a systematic review (published in the Journal of Vascular Surgery) of covered balloon-expandable stents for the management of aortoiliac occlusive disease. De Vries said: “Covered balloon-expandable stents are effective treatment options for aortoiliac occlusive disease as evidenced by the high rates of technical success and patency in all the stent grafts at one-year follow-up. They are preferred over bare metal stents for complex aortoiliac lesions, based on the documented five-year patency results for Advanta V12 in the COBEST trial.”
In the first talk, Jean-Paul de Vries, (Groningen, The Netherlands) reported the key findings from a systematic review (published in the Journal of Vascular Surgery) of covered balloon-expandable stents for the management of aortoiliac occlusive disease. De Vries said: “Covered balloon-expandable stents are effective treatment options for aortoiliac occlusive disease as evidenced by the high rates of technical success and patency in all the stent grafts at one-year follow-up. They are preferred over bare metal stents for complex aortoiliac lesions, based on the documented five-year patency results for Advanta V12 in the COBEST trial.”
Overall, the studies explored five different stents: Advanta V12 (Getinge), Viabahn VBX (Gore), Lifestream (BD), BeGraft (Bentley), and Jostent (Jomed).
De Vries said the rates of technical success and primary patency were both high, adding that that primary patency, at one year, “was 96.6% for Viabahn VBX and that is comparable to the Advanta V12 [83.6–96.4% at one year] and 94.4% for BeGraft [one-year data were not available for Lifestream or Jostent].” He further noted that five-year data were available for Advanta V12, showing a primary patency rate of 74.7% and a freedom from target lesion revascularisation rate of 67.4%.
During the discussion that followed his presentation, de Vries noted that comparative trials were required because, at present, the data are insufficient to provide “any proof that the new-generation devices perform better than the former generation ones”. However, he observed that three-year data for Viabahn VBX had recently been published and these were comparable to three-year results for Advanta V12.
“Excellent” three-year results with Viabahn VBX relate to stent graft’s unique design
Andrew Holden (Auckland, New Zealand), in his talk, further expanded on the three-year data from the VBX FLEX investigational device exemption (IDE) trial. The objective of the study, he explained, was “to evaluate the safety and efficacy of the VBX stent graft [Gore] for the treatment of arterial occlusive disease in patients with de novo or restenotic lesions in the common and/or external iliac arteries”. The primary outcome was a composite of major adverse events at nine months.
Overall, in VBX FLEX, 234 devices were implanted into 213 lesions (134 patients) across 27 sites. Holden said: “The Viabahn VBX is made up of a double polytetrafluoroethylene (PTFE) layer with independent stainless steel rings that give it flexibility and conformability.”
He reported that with the three-year data, freedom from target lesion revascularisation “was a very impressive 91.2%, which improves to 98.1% when we evaluate the clinically-driven target lesion revascularisation.” Furthermore, according to Holden, “a number of clinical parameters remained improved at three years,” including the Rutherford category, resting ankle brachial index, and walking impairment questionnaire scores. “In fact, of the patients who were at Rutherford 0 by three years, 80% had started from a baseline of Rutherford 3 or 4. Overall, 92% of patients improved at least one Rutherford category at three years,” he explained.
“The VBX FLEX clinical study is truly a real-world study, including complex aortoiliac occlusive disease (TASC II C and D lesions, 32.1%), that has shown excellent results. […] These are no doubt due to the unique design of this device,” Holden concluded.
The ensuing discussion emphasised that covered stents have become the first-line treatment for complex aortoiliac disease. “What we have seen, particularly in complex disease, is a real practice shift towards covered stents. I think one could consider using uncovered stents for very simple lesions, but obviously, for more complex cases, I think the standard of care is covered stents. Rupture of iliac arties is potentially catastrophic, but this is really very well managed by covered stents.”
Greenhalgh observed that there was a risk of rupture when implanting a covered stent because calcification meant that there was no elasticity in the artery. “When you start to expand it, surely, occasionally, there are ruptures? [Is the understanding that] the lining is going to take care of the leak?” he questioned.
Holden commented: “That is precisely right. As long as we are trying to optimise lumen diameter, we are going to see significant incidence of rupture. […] With covered stents, you are certainly much more comfortable being aggressive.”
Roger Greenhalgh (top left), Gunnar Tepe (top right), Rupert Bauersachs (bottom left), Thomas Zeller (bottom middle) and Joakim Nordanstig (bottom right)
Rivaroxaban reduces risk of comorbidities in patients with peripheral arterial disease
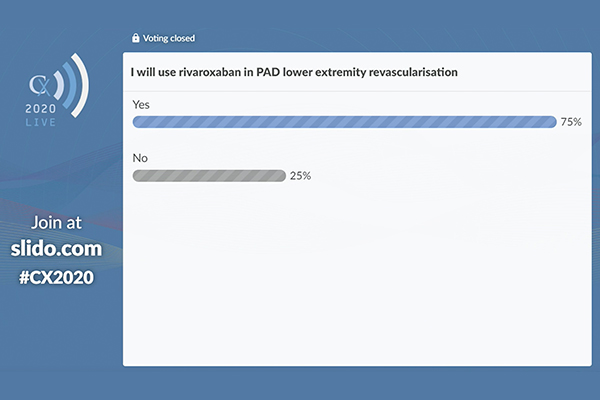
Three-quarters of the CX 2020 LIVE audience agreed with the statement: “I will use rivaroxaban in peripheral arterial disease lower extremity revascularisation”. Polling took place following a talk from Rupert Bauersachs (Darmstadt, Germany) giving the results of the VOYAGER PAD trial.
“After revascularisation, one in five peripheral arterial disease patients will suffer from acute limb ischaemia, major amputation, acute myocardial infarction, ischaemic stroke, or cardiovascular death within three years,” Bauersachs told CX 2020 LIVE delegates. The VOYAGER PAD trial, he went on to relate, therefore set out to evaluate the safety and efficacy of 2.5mg bid rivaroxaban plus aspirin versus aspirin alone to reduce thrombotic vascular events after peripheral revascularisation.
Bauersachs reported: “Rivaroxaban 2.5mg bid on top of aspirin was associated with a significant 15% reduction in major adverse limb events and major adverse cardiac events, with no significant increase in TIMI [thrombolysis in myocardial infarction] major bleeding. Acute limb ischaemia was significantly reduced by 33% compared to current standard of care.”
The study therefore met both its primary efficacy endpoint—a novel outcome measure combining major adverse limb events and major adverse cardiac events—and its primary safety endpoint—TIMI bleeding.
“Dual pathway inhibition treatment is the only antithrombotic regimen proven for significant benefit of the peripheral revascularisation in a large, randomised controlled trial,” Bauersachs concluded.
As questions began to flood in from Europe and Asia, it became clear that many pertained to Bauersachs’ rationale for using such a low dose of rivaroxaban. “It is a very low dose, 2.5mg,” Bauersachs agreed, adding, “the actual dose for treating venous thromboembolism, for example, is 30mg. Because it is such as low dose, it has to be given twice a day.
“As we know, these are high-risk patients, so the objective was, with a low dose of anticoagulants on top of antiplatelets, to reduce those outcomes.”
Responding to a question asking why a low dose of warfarin was not used, Bauersachs said that previous trials had shown “unacceptably high rates of bleeding” and had lacked efficacy.
Lastly, a CX 2020 LIVE viewer from Malaysia queried the recommended length of time for patients to take rivaroxaban plus aspirin, after revascularisation. In agreement with Tepe, Bauersachs said that it would be a lifelong treatment.
“I think the point we are talking about here,” Greenhalgh said in summary, “is that patients with that sort of peripheral arterial disease will have all of these other issues and comorbidities. It is not a standalone condition, it is a marker of arterial disease throughout the body’s vascular system; the more peripheral, the higher the likelihood it will be severe elsewhere in the body.
“So, the use of rivaroxaban enables the complications that come with this complex disease to be reduced, and comorbidities can be handled better.”
MIMICS-2 study three-year results: Swirling flow “is the answer”
Thomas Zeller (Bad Krozingen, Germany) presented the three-year results of a US IDE study, MIMICS-2, for which he is the European principal investigator. The prospective, single-arm, multicentre clinical trial found comparable efficacy and safety results for the helical BioMimics 3D stent (Veryan Medical) and drug-coated balloons or drug-eluting stents available on the market.
The primary endpoint for this study consisted of a safety endpoint, composite of deaths, major amputation, clinically-driven target lesion revascularisation through 30 days, and an effectiveness endpoint defined as primary patency at 12 months. The proportion of chronic total occlusions was 30%, and moderate-to-severe calcification was seen in 46% of lesions.
Both device and technical success was 100%, and the primary safety and primary efficacy endpoints were reached. Zeller told the audience that these results represent “a clear achievement” for the BioMimics 3D stent.
Three-year freedom from clinically-driven target lesion revascularisation was 81% for the entire patient cohort, and there was “only” a decrease by 8% of freedom from reintervention between one and three years, Zeller related.
Describing the device, Zeller said: “The BioMimics 3D stent represents a new bare metal stent, designed with a helical centreline, which imparts a nonplanar curvature to the stented femoropopliteal segment, inducing swirling flow.”
This mimics the natural flow of certain anatomy, Zeller explained. The vessel curvature in the aortic arch and the iliac arteries induces a swirling flow in normal anatomy, known to protect endothelial cells against atherosclerosis and restenosis, he noted. This is based on the lifetime work of Colin Caro (London, UK). Greenhalgh elaborated: “From what you have just told us, we can say Colin [Caro] was right, all along. Swirling flow is the answer.”
Zeller assessed the results of the BioMimics 3D stent from the MIMICS 2 study against those for contemporary devices, including bare metal stents, drug-coated balloons, and drug-eluting stents. “If you compare the BioMimics 3D stent results from the MIMICS 2 study with Supera (Abbott), Zilver PTX (Cook Medical), Eluvia (Boston Scientific), and the IN.PACT Admiral (Medtronic), we can see that freedom from reintervention up to three years is almost comparable,” Zeller said.
Zeller responded to a question from Tepe about the lower event rate seen in the long term, commenting: “It underlines the theory of the basal-protective and endothelial-protective effect of swirling flow. We have the acute phase, where we see the patency failures, which are induced by acute inflammation, resulting in neointimal hyperproliferation and, over time, it evens out regarding the need for reintervention. For me, the most interesting question would be: ‘What is the outcome of combining this sort of long-term protective stent design with acute suppression of neointimal hyperproliferation means with an antiproliferative agent?’ That would be a very interesting study to be seen in the future.”
Following Zeller’s presentation, Greenhalgh commented: “[The BioMimics 3D stent represents] a completely new way. We are hearing about the future here. This is not just practical reconstruction—you have just heard how the future is going to go.
“Caro spent his life analysing normal flow of an artery, and he believed that if the blood swirled, then it would reduce the risk of arterial disease, and we have just heard that that it is possible, and he was right. We have also had a peek into the future, about how the treatment might be.”
Absence of long-term benefit of revascularisation in claudicants, according to five-year data
The session also heard how a liberal strategy of revascularisation in patients with intermittent claudication resulted in a substantial improvement in health-related quality of life for two years, but this benefit was lost at five years. This is the conclusion presented by Joakim Nordanstig (Mölndal, Sweden), after he shared the five-year results from the IRONIC randomised trial.
The IRONIC trial investigators aimed to study the clinical effectiveness and cost-effectiveness of the two most widely offered treatment strategies for patients with intermittent claudication: lower limb revascularisation in combination with best medical treatment and exercise advice, versus best medical treatment and exercise advice only.
Speaking to the fact that the IRONIC trial included femoropopliteal lesions and aortoiliac lesions alike, Greenhalgh commented, “Assessors would say that the expectation of success or reconstruction over five years with the aortoiliac is far better than with the femoropopliteal. Some would argue that you should keep them separate because the expectation is different. They may present with intermittent claudication, so it is one presentation, but a different expectation of natural history.”
“I agree with that comment,” Nordanstig replied, “but for the purpose of this design, it was a real-world design.”
Asked if this would impact his clinical practice of patients with intermittent claudication going forward, Nordanstig said: “In a way, yes,” before elaborating. “It is a staged process, actually, and I try to really carefully inform every patient about the pros and cons with revascularisation. It is not a first line treatment for me, but I will re-evaluate all patients within a few months.”
The session was followed by an Industry Symposium supported by Concept Medical to explore where the field stands in terms of sirolimus devices, with a session titled “Overcoming the limitations of paclitaxel: A new sirolimus-coated balloon for the treatment of peripheral vascular disease”.









