Roadsaver – Carotid stenting with micromesh
The Roadsaver (Terumo), a self-expanding stent that has a dual layer micromesh design for sustained embolic protection, was used to treat a 73-year-old female with a high-grade symptomatic lesion in the internal carotid artery, Arne Schwindt (Munster, Germany) reported. He explained that she had an ipsilateral Amaurosis fugax eight weeks’ prior to the intervention and computed tomography (CT) angiography and Duplex confirmed a 80% right internal carotid artery stenosis. The patient also had arterial hypertension, hypercholesterolaemia, and type II diabetes.
Further to a having dual mesh design, Schwindt commented, the Roadsaver has “a flexible weave completely made of nitinol, an extremely flexible delivery system, and is retrievable and repositionable”. He explained that although accessing the lesion was the key challenge in the case, the risk of dislodging the stent’s guidewire system was low because of the system’s flexible delivery system.
According to the six-month follow-up result, the patient had no neurological events during the peri or post procedural period. Furthermore, convergent colour Doppler showed that the stent was patent with vmax of <100cm/seconds (previously, it was 190cm/sec). Schwindt concluded that the stent has “proven good deliverability, wall adaptation, and can be used with a variety of protection devices in challenging anatomies and lesions.”
Relay – Scalloped thoracic graft to the left subclavian artery and branched arch procedure
Mo Hamady and Joost van Herwaarden (London, UK, and Utretch, The Netherlands, respectively) jointly presented the case of a 43-year-old female patient with known hypertension who arrived at the hospital after she fell from steps at home. Because the patient presented with back pain, CT angiography was performed and this showed a non-traumatic saccular aneurysm of the thoracic aorta (ie. ductus diverticulum).
The presenters explained that the 9.5cm type 1 thoracic aortic aneurysm involved type III aortic arch, starting from the level of the innominate artery and extending to the distal thoracic aorta. There were more than 90 degree double bends at the distal end of the aneurysm.
According to van Herwaarden, the distance from the left subclavian artery to the aneurysm was too short for thoracic endovascular aortic repair (TEVAR) below the subclavian artery, and “further the ‘young’, highly curved aortic arch might have caused bird-beaking of a stent graft deployed in the aortic arch”.
Treatment options were TEVAR with covering of the left subclavian artery with or without revascularisation, TEVAR using a custom-made stent graft with a scallop for the left subclavian artery, or open repair.
The Relay (Bolton Medical) device, a custom-made branched thoracic stent graft with proximal scallop, was deployed in the aorta. Bridging stents to the innominate and left common carotid arteries were used to seal the aneurysm. The left common carotid to left subclavian artery bypass was used to revascularise the covered native subclavian artery.
Technical success was achieved and a postoperative CT angiography at one month showed exclusion of the aneurysm with open left subclavian artery and without bird-beak configuration.
Treatment of a chronic type B aortic dissection with Valiant Captivia after ascending aorta and arch surgery
Luigi Lovato (Bologna, Italy) presented the case of a 73-year-old female with a history of hypertension, ascending aorta surgery for aneurysm and chronic type B dissection from middle descending thoracic aorta. The patient was submitted to arch aneurysm surgical replacement. Serial CT imaging showed progressive severe dilation (>5mm/year) of aortic isthmus and middle thoracic aorta false lumen, joining 5.5cm at the level of the big entry tear with appearance of partial false lumen thrombosis.
The patient was considered for TEVAR of both aortic lesions. Lovato said that challenges arose due to the surgical reconstruction anatomy with angulation between the prosthesis and native aneurysmatic aorta and difficult visualisation of the reimplanted epiaortic vessel, creating a relatively short proximal neck, and discrepancy between the proximal and distal neck, because the sinusoidal intimal flap and the relatively distal entry tear forced them to cover the whole thoracic aorta extending next to the coeliac trunk where the true lumen is small.
He said that the Valiant Captivia (Medtronic) stent graft was selected because “it combines a solid capture tip system to obtain a high controlled stent deployment and low risk of wind-sock and bear-beck effect and, at the same time, an optimal hydrophilic delivery system to manage the advancement through a relatively small femoro-iliac access and tortuous aorta”.
During the procedure, Lovato explained, they adopted a different approach, delivering the distal stent graft first to solve the neck discrepancy. The second stent was deployed afterwards, proximally, in the surgical prosthesis and with the distal part inside the first distal stent. The sizing was sorted to achieve the best oversizing in the prosthesis, avoiding type I/III endoleak.
The pre-discharge CT control (six days later) showed an optimal stent graft positioning without type I/III endoleak. A small type II endoleak was visible at isthmic level. There was a moderate retrograde flow from the abdominal false lumen reaching the middle thoracic aorta, but only in the late CT scan.
“The case demonstrates the need for careful planning to meet the anatomical challenges in patients with previous arch surgical reconstructions and the feasibility and safety of this ‘distal first approach’ of TEVAR for type B dissection. This technique can solve the frequent neck discrepancy of these patients avoiding the severe complication of distal false lumen aneurysmal degeneration, caused by the intimal flap rupture induced by the stent graft radial forces,” Lovato concluded.
ZENITH Alpha thoracic endograft
Geert Schurink (Maastricht, The Netherlands) reviewed the case of an 80-year-old female whose thoracic aneurysm was discovered after she underwent a computed tomography (CT) scan for a deep vein thrombosis. He said she was “a very fit” 80-year-old but did have a kyphosis and moderate femoral pulses.
The patient underwent TEVAR with the Zenith Alpha thoracic endograft (Cook Medical). Afterwards, in the recovery unit, mean arterial pressure was >85mmHg and a cerebral spinal fluid drain was not put in because of her concerns about her kyphosis. However, two days after the procedure, she lost sensation in both lower legs and experienced weakness in her left leg. A cerebral spinal fluid drain was put in and her mean arterial pressure was >90mmHg. Schurink reported that the patient made an “almost complete recovery”. He added that he did not believe the loss of sensation was a result of an embolization (in the distal aorta) because if it had been, he said, she would not have made a recovery. Furthermore, there were no signs of emboli on angiography.
After the case was presented, the audience not only discussed when to put in a cerebral spinal drain after TEVAR but also reviewed whether patients were allowed to get too hypotensive compared with their relative baseline pressure.
Nellix – Endovascular aneurysm sealing (EVAS) with polymer in the aortic sac
In the case that Michel Reijnen (Arnhem, The Netherlands) reported, a 70-year-old male patient, with a cardiopulmonary history, presented with an infrarenal aneurysm with a maximum diameter of 55mm.
The patient was treated using the Nellix endovascular aneurysm sealing system (Endologix). Reijnen explained that Nellix consists of two balloon-expandable endoframes surrounded by endobags that are filled in situ with a biostable polyethylene glycol (PEG)-based polymer to completely fill the aneurysm sac, thus sealing the entire aneurysm.
The case showed that the infrarenal neck diameter and length were respectively 28mm and 35mm. Access was adequate and the calculated volume to seal the aneurysm was 64ml. After a bilateral cutdown, an angiography was performed with a calibrated catheter. Subsequently, two Nellix endosystems were inserted, positioned and deployed. A prefill with saline solution, to a pressure of 180mmHg, was performed showing a complete exclusion of the aneurysm. The saline solution was then replaced by 55mL of polymer to reach a final pressure of 190mmHg. During curing the Nellix balloons were re-inflated.
Completion angiography showed a complete exclusion of the aneurysm with patent renal and hypogastric arteries. Throughout one year follow-up, there were no complications.
Reijnen advised that the “key points for success are case planning and extensive knowledge of the device and technique, which differs from regular EVAR”.
The INCRAFT low profile device introduced percutaneously
A patient with a 55mm aortic abdominal aneurysm was the subject of the case that Hany Zayed, Gianbattista Parlani and Jost Philipp Schäfer (London, UK; Perugia, Italy; and Kiel, Germany, respectively) presented. They reported on how the patient was treated with an Incraft endograft (Cordis).
The procedure was performed under local anaesthesia with a bilateral percutaneous approach. The presented case showed the endograft’s main body delivery and the possibility to reposition the graft, as well as the Iliac limbs implantation accommodating the device into the broad spectrum of maximum and minimum overlapping in order to obtain the correct length of the device to preserve the hypogastric artery.
The completion angiography performed at the end of the procedure showed precise endograft placement with the start of the covered portion of the device immediately below the lowest renal artery and total coverage of both common iliac arteries with preservation of the hypogastric artery.
Parlani noted that the key message of the presentation “is that this ultra-low profile device is designed for proximal and distal placement accuracy and allows for customisation during the procedure to accommodate a wide range of anatomical sizes. This broad anatomical coverage is offered with a minimal number of product codes for easier pre-procedural planning”.
Challenging AAA anatomy treated by Endurant IIs stent graft
Medtronic’s Endurant IIs stent graft has several advantages and benefits for managing patients with challenging anatomy, Johannes Gahlen (Ludwigsburg, Germany) said in his case presentation of a 64-year-old male with an abdominal aortic aneurysm of 5.2cm.
He explained that the device has expanded anatomical customised options, including a 20% reduction in distal diameter compared with select Endurant II bifurcation stent grafts, in-situ sizing with select ipsiliateral limbs, allowing a 3–5 stent overlap for adjustment curing cases, and easier pre-case planning to simplify sizing. It has also has a “better anatomical fit” with “less material encumbrance in tight aortic bifurcations and “better precision in distal landing to avoid type IIb endoleak or unintended coverage of the hypogastric artery.
Gahlen reported that, in the case he was presenting, endovascular aortic aneurysm repair was performed with the graft using the three-piece approach. He described the patient’s anatomy as challenging because there was kinking and stenosis in the left common iliac artery and the right common iliac artery was dilated.
According to Gahlen, the usual practice is to perform the follow-up CT scan at six months, but three-month follow-up was planned for this patient because the device was relatively new.
The Aorfix endovascular stent graft
The patient in the case presentation from Robert Beasley (Miami, USA) was an 81-year-old male with a history of diabetes, hypertension, chronic obstructive pulmonary disease, coronary artery disease, and smoking (he quit in 2009). Beasley commented that the patient was found to have a 7.8 x 7.3 x 10cm abdominal aortic aneurysm with an angle of
He explained that the patient was treated with an Aorfix endovascular stent graft (Lombard). Features of this device include, according to Beasley, radioplaque markers on the stent body graft and legs for “precise positioning”, 8mm sealing zones at proximal distal ends of stent grafts, and a “fishmouth” fixation mechanism (the proximal end of the graft expands into a fishmouth shape). Beasley called this mechanism a “unique design concept” that allows the endograft to be placed transrenally and has four pairs of hooks to “resist migration forces”.
The E-liac device in complex iliac aneurysm
In his presentation, Jost Philipp Schaefer (Kiel, Germany) explained that a 56-year-old male patient developed a dissected iliac aneurysm in the left common iliac artery after undergoing surgical repair of an aortoiliac aneurysm with an aortofemoral (right) and aortoiliac (left) graft interposition.
The dissection caused a severe stenosis at the ostium of the internal iliac artery with symptoms of buttock claudication and erectile dysfunction. Via combined left groin and transbrachial approaches, the dissected iliac aneurysm is repaired by utilising the E-liac device and an E-ventus stent graft (Jotec) as bridging stent. The applicable techniques were demonstrated step-by-step, including the completely percutaneous approach under local anaesthesia, the successive deployment of the iliac-side-branch graft, and the placement of the bridging stent graft.
Schäfer said that this Edited Live Case “demonstrates the technique of endovascular repair of iliac artery aneurysm with the new E-liac device. Available off-the-shelf, the E-liac device is a low-profile iliac-side-branch graft, offering a percutaneous treatment option for a wide range of aortoiliac aneurysmatic pathologies”.
Control of infection with prosthetic grafts: added value of INTEGARD SYNERGY
Jean-Paul de Vries (Nieuwegein, The Netherlands), in his case presentation, explained how the Intergard Synergy (Maquet) graft was used to treat a 62-year-old female with (as identified on CT scan) a mycotic abdominal aorta combined with Leriche syndrome. He explained that the graft was the only one to be loaded with two different antiseptics, adding that these antiseptics (silver acetate and triclosan) are “not limited to one target and do not have the likelihood to produce bacterial resistance”. De Vries added that the graft was “designed for routine prophylactic use” and was not associated with any contraindications for contaminated implant sites.
In this case, the patient was on the hospital ward for 12 days after the operation and received penicillin for eight weeks afterwards. According to de Vries, there was no sign of infection at the two month postoperative CT scan.










 Chair Roger Greenhalgh (top left), moderator Andrew Holden (top right), Sapan Desai (bottom left), Sabine Steiner (bottom middle), and Misti Malone (bottom right)
Chair Roger Greenhalgh (top left), moderator Andrew Holden (top right), Sapan Desai (bottom left), Sabine Steiner (bottom middle), and Misti Malone (bottom right) 




 Andrew Holden
Andrew Holden 































 Fadi Taher
Fadi Taher 


































 Lowell Kabnick
Lowell Kabnick 













 Ross Naylor
Ross Naylor 
 Stephen Black
Stephen Black 
 Mark Whiteley
Mark Whiteley 


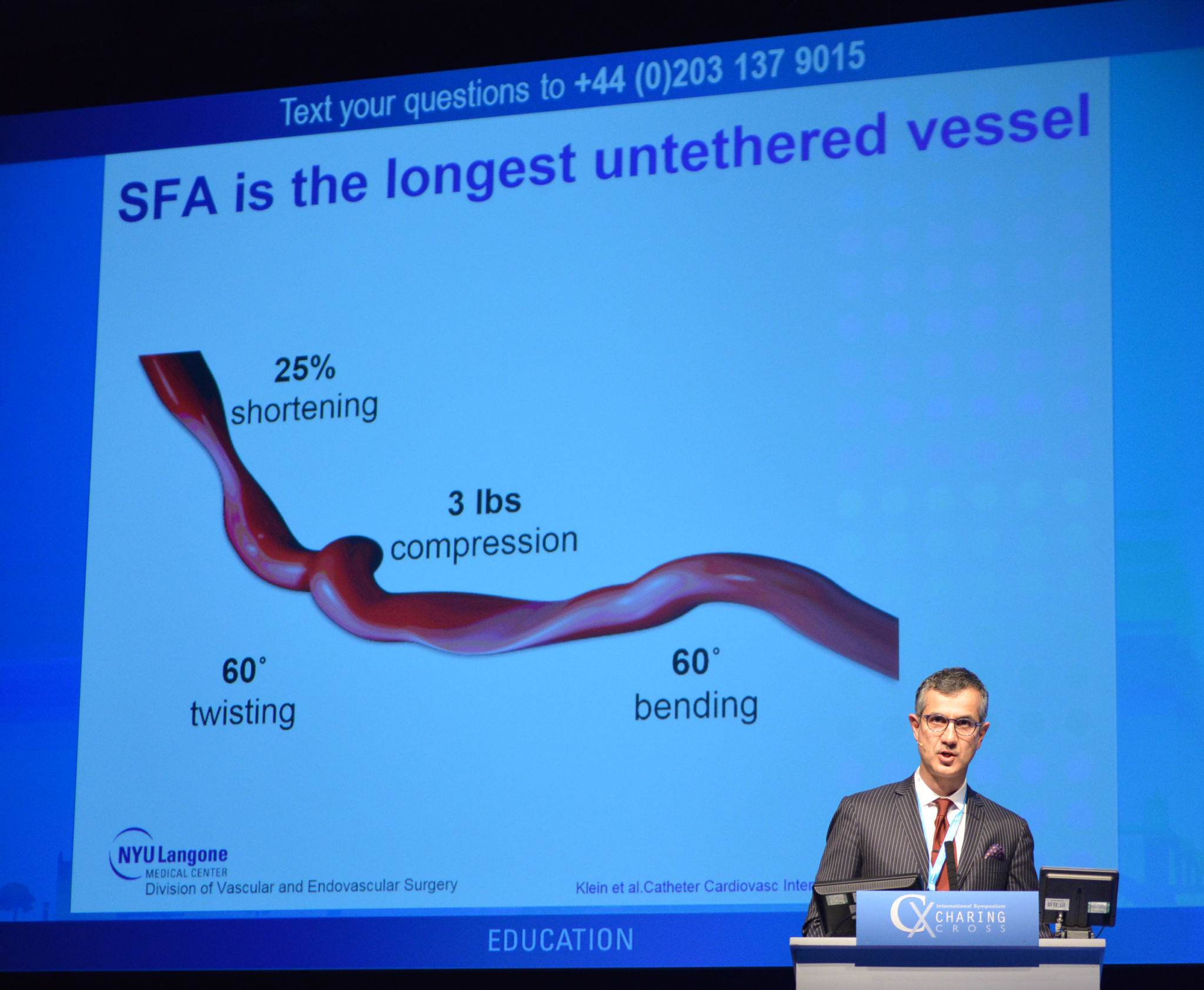
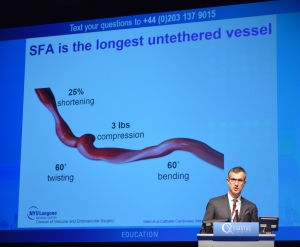

 CX 2015 Peripheral Arterial Live Cases.
CX 2015 Peripheral Arterial Live Cases. 
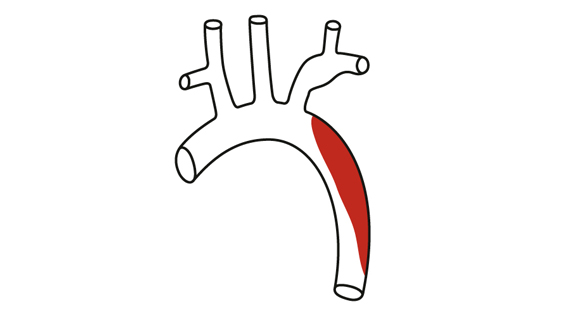
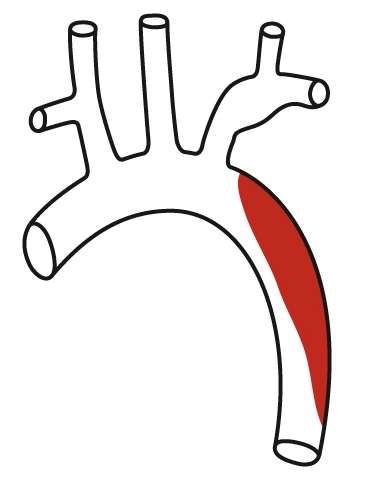 Type B dissection.
Type B dissection. 

 Roger Greenhalgh.
Roger Greenhalgh.